MODULE 1: Overview of Patient Support Programs
OBJECTIVES
After reading this module, you should be able to:
- Describe the key elements of Patient Support Programs in Canada.
- Describe the different types of Patient Support Programs.

In this module, we will review the different elements of a Patient Support Program and describe how these different services could support a patient like Angelo. By the end of this module the following questions will have been addressed:
- What is a PSP?
- Why do we have PSPs?
- How long have PSPs been used in Canada?
- Who operates PSPs?
- What services do PSPs offer?
- What are the different types of PSPs?
- How do PSPs vary in size?
- What is the Lifecycle of a PSP?
What is a Patient Support Program?
A Patient Support Program (PSP) is a privately-operated set of services provided by pharmaceutical and biotech manufacturers to support a patient through a number of therapy-related needs.
These programs are designed to assist patients and healthcare practitioners (HCPs) with services that can help reduce the barriers to taking the medication. These services can include nursing services, reimbursement navigation, infusion services, education, pharmacy services, financial assistance and other supports (e.g. psychosocial services).
 Why do we have PSPs in Canada?
Why do we have PSPs in Canada?
In Canada, not all healthcare services are covered or paid for within our National Healthcare System. In particular the cost of medications. Depending on the province you live in and/or the type of disease you have, medications may not be covered if you are over 65 or under 25. Some people have private health insurance through their employer which can assist in covering some of their medication costs.
What are PSPs not intended to do?
When considering Patient Support Programs, it is also important to be aware that these programs should not be designed to:
- Generate safety and efficacy data
- Provide medical advice or treatment suggestions to patients
- Modify a physician’s recommended course of therapy
How long have PSPs been used in Canada?
Patient Support Programs have been operated in Canada for over 20 years; more so in the past 5 years. With the rise in numbers of specialty and biologic products and higher costs of these medications, the number of PSPs has grown significantly. While there was not an official list of PSPs in Canada at the time of writing this course, it is estimated that there were well over 250 programs being operated.
Who operates PSPs?
In Canada, there are several third party providers (Providers) who administer and operate these programs. Most Provider-run programs offer specialty pharmacy and nursing services. Occasionally, some pharmaceutical or biotech companies (Manufacturers) manage their own PSPs or a portion of their PSPs without the support of external Providers.

What services do PSPs offer?
PSPs offer a wide range of services aimed at assisting patients with access to the medications they require. More details on these services will follow in future modules.
What are the different types of PSPs?
The term Patient Support Program is a broad classification of a collection of patient services. These programs vary on how they are designed and structured. Two primary
ways that PSPs can be sub-classified are by the type of Hub or Service Model used.
PSP Hubs
A PSP Hub is a description of an operational structure of a program.
There are three common types of PSP Hubs:
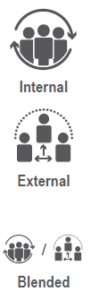 Internal Hub – the Manufacturer runs their own program using their Full-time Employees
Internal Hub – the Manufacturer runs their own program using their Full-time Employees
External Hub – a third-party Provider runs the Manufacturer’s program using staff employed by the Provider
Blended Hub – a mix of both Manufacturer and Provider run services working in conjunction e.g. a Manufacturer’s Program Manager may oversee the program from a strategic oversight role, however, the day-to-day operations are managed by the external Provider’s Program Manager
Companies determine which Hub structure to use based on several criteria such as the number of patients in the program, the required geographic reach, and the types of services being offered. Most PSPs in Canada are designed as External Hubs.
Service Models
There are generally two types of service models used in PSP sub-classifications:

Full-time Equivalent (FTE) – these programs are based on the number of full-time employees utilized to provide the full range of program services. An example of a FTE is a Nurse Case Manager (NCM), or a Single Point of Contact (SPOC). The more FTEs used in a program, the more expensive the program may become. The FTE service model is considered a fixed cost approach.
Fee for Service (FFS) – these types of programs are based on a discreet fee for service/variable cost approach, meaning each time a program service is utilized, a fee is charged back to the Manufacturer by the Provider. Common services used in FFS models are co-pay card programs, on-demand nursing support, drug supply fulfillment and other innovative patient tools and resources such as portals.
 It is important to note that some larger PSPs may use a mix of both FTE and FFS models to execute their programs.
It is important to note that some larger PSPs may use a mix of both FTE and FFS models to execute their programs.
How do PSPs vary in Size?
PSPs can vary in size based on three perspectives:
- number of staff on the PSP team
- number of patients enrolled in the program
- number of products in a program
Size of the PSP Team
Each program will be supported by a Manufacturer PSP team regardless of who ultimately operates the program. PSP teams can range in size from no team, less than one person, to a large team of 20 or more staff. Most companies have PSP Teams with 1-3 Program Managers. The size of a team is generally related to the number of PSPs the company is operating overall.

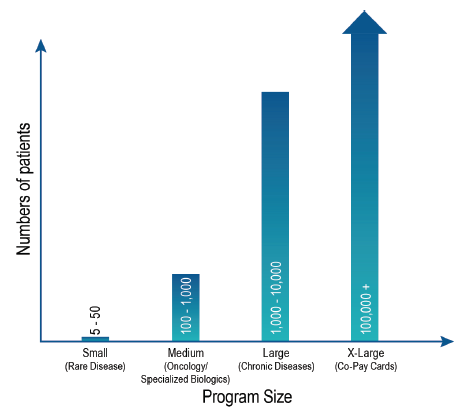
Numbers of Patients
The number of patients in Canadian PSPs can significantly vary. Some programs may have as few as 5 patients and some will have over 100,000 patients. The number of patients within a program is largely related to the incidence and prevalence of the disease being treated by the medication in the PSP. In the case of PSPs offering co-pay cards, the total number of patients can be extensive.
Number of Products in the PSP
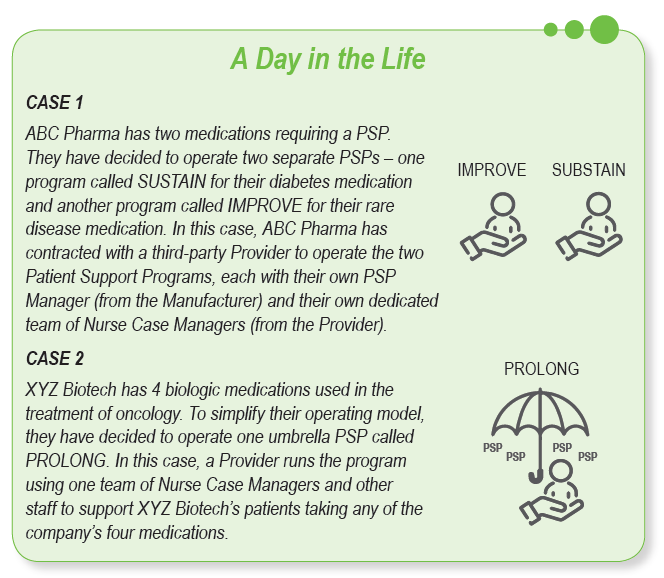
Most Patient Support Programs service a single product. However, some Manufacturers prefer to use Umbrella PSPs that involve several products residing under one program with a common name and brand. It is important to note that Umbrella PSPs could be run by one PSP Manager or separate PSP Managers depending on the number of patients in each of the programs.
What is the Lifecycle of a PSP?
As with any initiative, Patient Support Programs follow a typical staged approach to start-up, implementation and closure. This phasing is referred to as the PSP Lifecycle which has five stages:
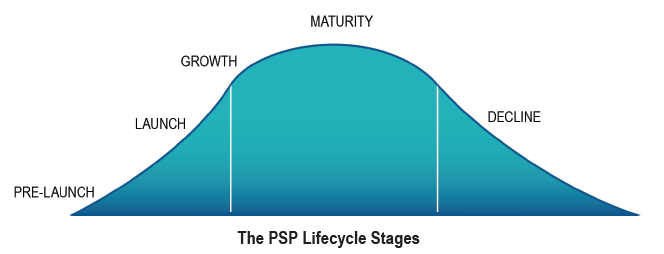
PRE-LAUNCH
The pre-launch phase usually begins 12 to 18 months prior to launch, although in some cases, companies may only have 6 months pre-launch to prepare. During this stage, the Manufacturer’s Executives in partnership with the commercial and medical teams plan the design and implementation elements of the program.
Key areas of focus during pre-launch may include:
Patient Access Challenges – identifying and prioritizing patient issues relative to the distribution and use of the new medication.
Program Design – determining what type of program is required to efficiently and effectively optimize patient access. Also, reviewing corporate SOPs and Guidance documents to ensure governance throughout the process.
Budget Forecasting – determining the funding available to run the PSP and identifying scenarios that could impact the program’s costs over time.
Request for Proposal – preparing a structured RFP process and selecting a Provider.
Special Considerations – assessing if there are any unique considerations to plan for e.g. is there a need for a Special Access Program?
Transferring Patients Approach – partnering with the medical team and HCPs to determine the best approach for transferring patients from clinical trials and/or special access programs to the new program.
Developing & approving the PSP components – ensuring design and development of several of the program’s operational processes and tools such as the enrolment form, AE reporting, data reporting, welcome kits, etc.
Preparing internal & external organizations for launch – creating and executing implementation plans to ensure a seamless and efficient program launch such as communications, training and team integration.
LAUNCH
Once the PSP is ready to launch, a new series of activities commence. This launch phase usually occurs one to two years post program initiation. The PSP Launch phase is very important to a product’s overall introduction as it assists both patients and physicians with quicker access to the new therapy. When a new product is launched in Biologic/Speciality Medicine, the HCPs first question to the company is often, “Do you have a PSP?” as they want to ensure their patients can access the medication without a large financial burden.
There are generally two stages of activity during this phase:
Pre-Public Reimbursement (Year One)
When a product is first launched, there is often minimal/no Public Insurance coverage leaving the patient to take-on a significant financial burden. PSPs can support HCPs and their patients without coverage during the first year of product availability by providing access to free medications while they wait for coverage approvals. Ways that PSPs provide this access support include:
- Transfer of Clinical Trial or Special Access Program patients
- Compassionate Use
- Bridging
- Private Insurance Assistance
Post-Public Reimbursement (Year Two and beyond)
Post-Reimbursement support begins when the Public plans begin providing approvals and paying for medications. PSPs seek to support patients with financial assistance during this time, ensuring they have access to their medications until coverage can be secured. The programs are able to support through services such as:
- Transitioning Bridging Patients to Commercial Patients
- Planning support for “No Reimbursements” or Cash-paying patients who fall through
the gaps in provincial and/or private reimbursement coverage - Co-pay assistance
- Reimbursement navigation and financial assistance
GROWTH
As the program matures, there will be instances where the PSP team and their Provider partners will need to plan for additions and/or changes to the program. This may result in a growth of or enhancements to the initial PSP. This growth phase may be triggered as a result of:
- New product indications (the PSP returns to the Launch phase for each new indication)
- The need for additional New Services
- Continued or new requests for Bridging and Compassionate support
- Annual Reviews
MATURITY
The PSP reaches the Maturity stage when the product it supports is in a similar stage in its product lifecycle. Key activities undertaken during this phase include:
- Patient Enrolment reviews (assessing patterns – steady, increasing, decreasing)
- Budget allocation and decision-making (does the program need to maintain the same level of financial support compared to earlier stages?)
- Current services reviews – what services are still required and what services are no longer required?
- Adherence support – assist long-term patients to maintain their drug therapy schedule
DECLINE
The Decline stage occurs when most patients begin leaving the program and/or when a brand’s budget no longer supports the provision of a PSP. During this final stage, PSP Managers/Teams partner with their Providers to ensure:
- Final financial/budget planning and reconciliation
- Program Services debriefs – how well did the program meet its objectives?
- Determine if alternative Financial Assistance Programs are needed
Why is it important to manage the PSP Lifecycle?
There are a number of reasons that compel Manufacturers to proactively manage the Lifecycle of their PSP program(s):
 Program Costs – as the number of patients within the program increases Manufacturers may need to look at their Program Model i.e. transition from FFS to FTE model.
Program Costs – as the number of patients within the program increases Manufacturers may need to look at their Program Model i.e. transition from FFS to FTE model.
Patient Services – as products mature in their lifecycle the need for Financial Support may change.
New Patient Access Challenges – new challenges may emerge with financial impact that may need additional Program services and support.
New Market Entrant Challenges – new products may be launched that impact program enrolment – patients transitioning to newer therapies and/or switching between programs.
Program Design based on Product Lifecycle Stage – as the product moves to the Patient Withdrawal phase and patient enrolments drop, Manufacturers may shift back to an FFS model to conserve costs.
MODULE RECAP
- A Patient Support Program (PSP) is a group of services provided to help reduce barriers to patients requiring a particular medication.
- Examples of program services include nursing services, reimbursement navigation support, infusion and injection services, patient education, pharmacy services, and financial support services.
- PSPs are provided to patients by pharmaceutical and biotech Manufacturers (Manufacturers) for medications with unique requirements related to distribution and patient support.
- In Canada, there are several third-party providers (Providers) who administer these programs.
- Patient Support Programs can be sub-classified based on the type of structural hub approach or the type of service model used.
- PSPs can also vary in size relative to the number of patients within the program, products & services in the program or the size of the PSP Team.
- Patient Support Programs progress through a PSP Lifecycle that guides the startup, implementation and closure of the program. The PSP Lifecycle has five stages.
KNOWLEDGE CHECK
- What is a PSP?
______________________________________________
______________________________________________
______________________________________________ - What are the 3 types of hubs used to structure PSPs?
______________________________________________
______________________________________________
______________________________________________ - What are the 2 service model types using in PSPs?
______________________________________________
______________________________________________
______________________________________________ - What 3 ways do PSPs vary in size?
______________________________________________
______________________________________________
______________________________________________ - List the PSP Lifecycle stages.
______________________________________________
______________________________________________
______________________________________________ - Which Lifecycle stage is the most important and why?
______________________________________________
LEARNING JOURNAL
Areas I would like to further explore are:
- ______________________________________________
- ______________________________________________
- ______________________________________________
